The HXP056 is an oral small-molecule drug candidate for the treatment of retinal diseases, intended for conditions such as wet age-related macular degeneration (wAMD), diabetic macular edema (DME), and retinal vein occlusion (RVO). This small molecule is designed to overcome the significant and well-known technical challenge in the field: achieving sufficient penetration of the blood-retinal barrier (BRB) to reach the affected fundus tissue, while optimizing systemic exposure to ensure patient safety. It has the potential to become the world's first oral drug for treating these fundus diseases, representing a groundbreaking innovation with technological disruptive potential and immense commercial viability in the global market. Currently, all available retinal disease treatments are administered via intravitreal injections, a method that is not only highly inconvenient but also carries risks of infection and retinal damage, making long-term adherence difficult for patients. The HXP056 project innovatively develops an oral therapeutic approach for retinal diseases, addressing a significant unmet clinical need and potentially enabling more patients to manage retinal diseases from the comfort of their home.
Preclinical studies have demonstrated that this small molecule can penetrate the blood-retinal barrier (BRB) in mice and rats, exhibiting high and sustained distribution in ocular tissues with a long half-life, while being rapidly metabolized and cleared from systemic circulation without accumulation in other organs. This property enables it to exert localized therapeutic effects within the eye while mitigating potential safety concerns associated with systemic exposure. In the hVEGFA-transgenic mouse model, this small molecule significantly inhibited fundus vascular leakage, demonstrating a dose-dependent efficacy significantly superior to that of Aflibercept.
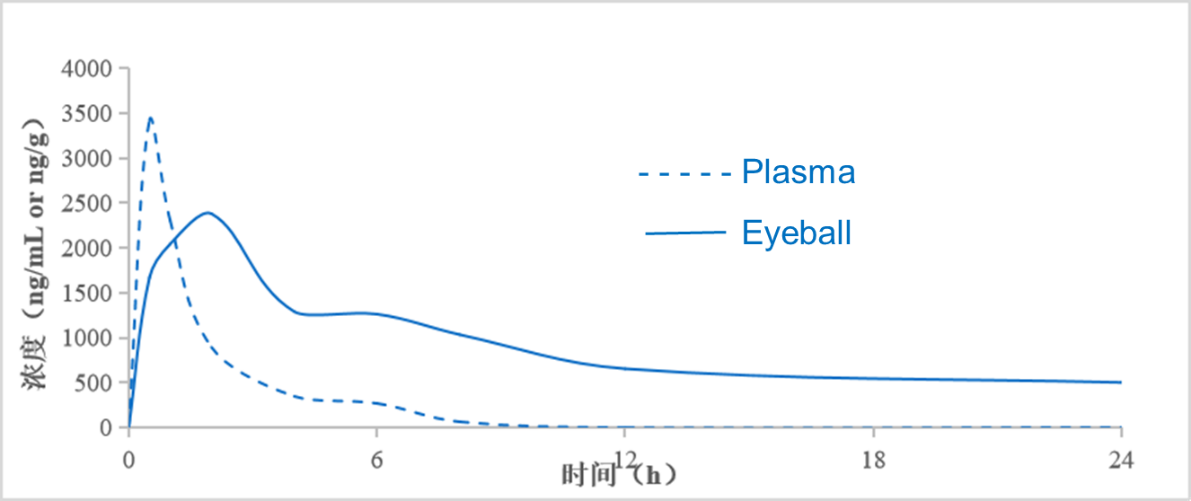
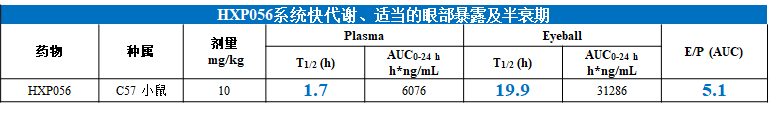
Figure 1: PK Exposure of HXP056 in the Blood and Eyes of C57 Mice
- ●Rapid systemic metabolism, short half-life, and no organ accumulation, reducing toxicity associated with systemic exposure.
- ●High ocular distribution, long half-life, exerting localized efficacy.
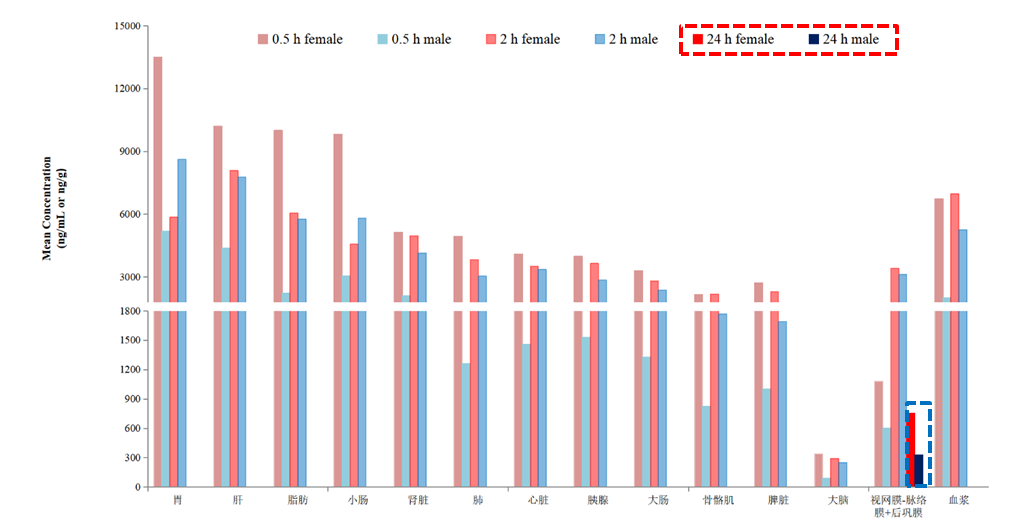
Figure 2: Distribution of HXP056 in the Fundus and Other Tissues of BN Rats
Following a single oral gavage dose of 10 mg/kg HXP056 in BN rats, the drug was rapidly distributed. At 24 hours post-dose:
●HXP056 concentrations in all organ tissues and plasma were below the lower limit of quantitation.
●HXP056 concentration in the retina-choroid + posterior sclera remained at approximately 15% of its peak concentration.
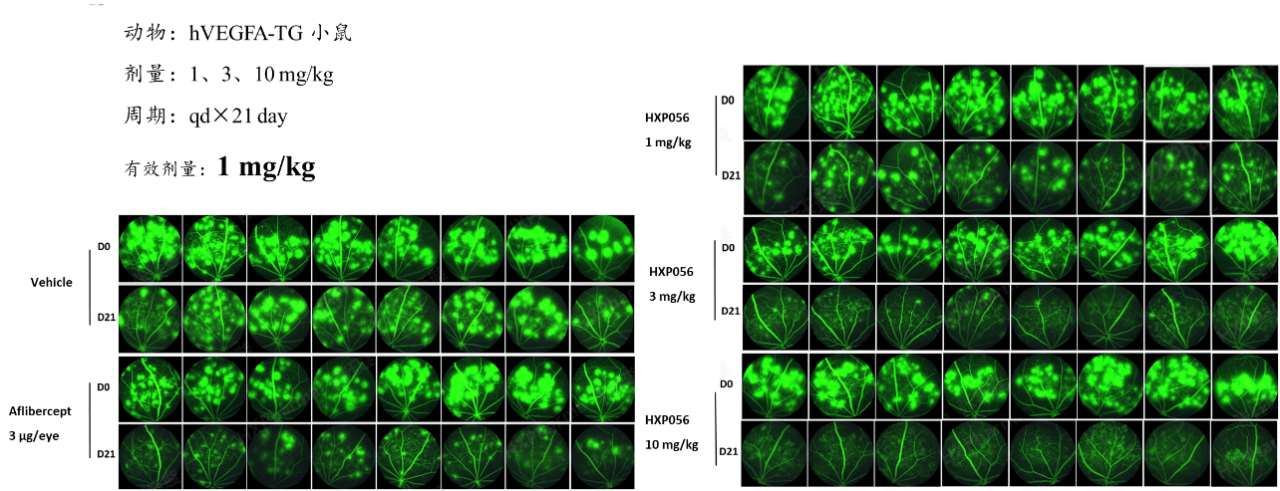
Figure 3: Efficacy of HXP056 in the hVEGFA-Transgenic Mouse Model
HXP056 significantly inhibited fundus vascular leakage in the hVEGFA-transgenic mouse model at doses of 1, 3, and 10 mpk.


